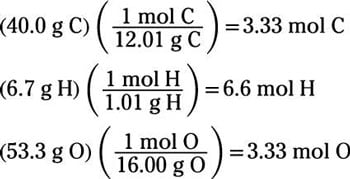
As multiple compounds can have the same lowest whole number ratio of elements - for example CH 2 is the empirical formula for both C 2 H. The empirical formula is the lowest whole number ratio of elements in a compound. Learn more about our school licenses here. The percent composition of a compound is often experimentally determined and used to derive the compound’s empirical formula. We’ll give you challenging practice questions to help you achieve mastery of AP® Chemistry.Īre you a teacher or administrator interested in boosting AP® Chemistry student outcomes? Albert.io lets you customize your learning experience to target practice where you need the most help. You can also find thousands of practice questions on Albert.io. Good luck and happy studying! Looking for AP® Chemistry practice?Ĭheck out our other articles on AP® Chemistry. It is convenient to consider 1 mol of C 9 H 8 O 4 and use its molar mass (180.159 g/mole, determined from the chemical formula) to calculate the percentages of each of its elements: C 9molC × molarmassC molarmassC 9H 18O 4 × 100 9 × 12.01g / mol180.159 g / mol × 100 108.09g / mol 180.159g / mol × 100 C 60.00C. That’s all you need to know about how to calculate an empirical formula for the AP® Chemistry exam. So, we calculate an empirical formula of CH_3! Likewise, 1.0 mole of H 2 O is composed of 2.0 moles of hydrogen and 1.0 mole of oxygen. Thus, H 2 O is composed of two atoms of hydrogen and 1 atom of oxygen. The ratios hold true on the molar level as well. Since we don’t know the mass of the sample, we can just simply assume a weight of 100 \text of hydrogen. An empirical formula tells us the relative ratios of different atoms in a compound. We need to have mass of the two elements in grams in order to calculate an empirical formula. 
For example, let’s say that you have a compound composed of nitrogen and oxygen, where nitrogen makes up 30.4% of the compound by mass. Figure Out How Much of Each Element is in the Compound.ĭepending on the question, this step can take different forms. You’ll learn how to make these calculations in this section of the AP® Chemistry Crash Course. Naturally, the AP® Chemistry exam is almost guaranteed to ask you to calculate an empirical formula for at least one substance, if not more. Though it’s not quite as useful as a molecular formula, an empirical formula still tells you a lot about the substance, and it’s possible to calculate it with much less information.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
